23歳女ですが、水虫です・・・。誰にも言えないくらい恥ずかしく、でもそのままにもしておきたくないのでこれを買いました。はじめて局留めで受け取りましたが、難しくなかったです。使って3日になりますが、状態は良くなってて、痒さはほとんどありません。

左記クレジットカード、銀行振込、コンビニ決済に対応





更新日:2025/6/3
ケトクリームは、白癬菌による水虫、いんきんたむしなどに効果的な抗真菌薬です。
また、マラセチアによる脂漏性皮膚炎に対しても効果を発揮します。
| メーカー | メッド・マナー・オーガニクス(Med Manor Organics) |
|---|---|
| 有効成分 | ケトコナゾール |
| 効果 | 白癬(水虫)・その他皮膚真菌症の改善 |
| 副作用 | 塗布部位の灼熱感やかゆみなど |
| 用法 | 1日1~2回、患部へ塗布 |
ケトコナゾールには、白癬菌やマラセチアといった真菌の細胞膜を構成するエルゴステロールの合成を阻害する作用があります。この作用によって真菌の増殖を抑え、症状を緩和させます。
ケトクリーム15gに、ケトコナゾール2%が配合されています。

| 個数 | 販売価格(1本あたり) | 販売価格(箱) | ポイント | 購入 |
|---|---|---|---|---|
| 1本 | 2,960円 | 2,960円 | 88pt | |
| 5本 | 1,792円 | 8,960円 | 268pt | |
| 10本 | 1,376円 | 13,760円 | 412pt |






①1万円以上で送料無料
1回の注文で10,000円以上だった場合、1,000円の送料が無料となります。
まとめ買いをすると1商品あたりのコストパフォーマンスが高くなるためおすすめです。
②プライバシー守る安心梱包
外箱に当サイト名や商品名が記載されることはないため、ご家族や配達員など第三者に内容を知られることは御座いません。

③100%メーカー正規品取り扱い
当サイトの商品は100%メーカー正規品となっており、第三者機関による鑑定も行っております。
商品の破損などがあった場合は再配送などにて対応させて頂きますので、ご連絡頂ければ幸いです。

④いつでも購入可能 処方箋不要
サイト上では24時間いつでもご注文を受けております。
また、お電話によるご注文も受け付けておりますのでネットが苦手な方はお気軽にどうぞ。

⑤商品到着100%
商品発送後はお荷物の追跡状況が分かる追跡番号をご案内させて頂きます。
郵便局には保管期限がありますのでご注意ください。
・自宅配達で不在だった場合の保管期限・・・16日間前後
・郵便局留めとした場合の保管期限・・・7~30日間

⑥コンビニ決済利用可能
ご近所のコンビニにていつでもお支払可能です。
セブンイレブンに限り店舗での機械操作を必要とせず、手続き完了後に表示されるバーコードや払込票番号をレジに提示することでお支払い頂けます。

ケトクリーム 30g x 1本
2,960円
ポイント:88pt
10,000円以上購入で送料無料
在庫あり

23歳女ですが、水虫です・・・。誰にも言えないくらい恥ずかしく、でもそのままにもしておきたくないのでこれを買いました。はじめて局留めで受け取りましたが、難しくなかったです。使って3日になりますが、状態は良くなってて、痒さはほとんどありません。
カンジダになったとき、何度か使ったことがありますが、綺麗に治るかわりにやや副作用が出る。一瞬カンジダが悪化したのかと不安になってしまいました。
脂漏性皮膚炎に対しても効果があります。脂漏性皮膚炎は皮脂の多い部位に起こる炎症ですが、原因の一つである真菌に働きかけることで、症状を和らげることができます。症状に応じて1日2回塗る方法が推奨されていますので、使用方法に注意しましょう。
症状の改善には通常2〜4週間程度かかることがあります。特に脂漏性皮膚炎の場合は、1日2回の使用で早期改善が期待できるとされていますので、根気よく続けることが大切です。
ケトクリームは水虫(足白癬)にも効果があります。水虫は皮膚の真菌感染によるもので、クリームを1日1回患部に塗ることで、菌の増殖を防ぎ、症状を改善することができます。毎日続けて塗ることが治療成功のカギになります。
カンジダが原因となる皮膚トラブルに効果を発揮します。皮膚カンジダ症の患部に塗ることで、原因となる真菌の増殖を抑えて、症状を改善してくれます。使い続けることでしっかりとした効果が期待できるので、焦らず継続しましょう。
白癬やカンジダ症、癜風の場合は1日1回、脂漏性皮膚炎の場合は1日2回、患部に塗るのが基本です。症状や種類によって使い方が異なるので、医師や薬剤師の指示に従って使いましょう。毎日の使用を続けることで、しっかりと効果を発揮します。
ケトクリームは、患部を薄く覆うくらいの量を塗るのが目安です。塗りすぎる必要はなく、適量をやさしく広げるだけで十分な効果が期待できます。使用量に迷ったときは、薬剤師に相談してみると安心です。
目の周りや粘膜には使わないように注意してください。また、傷口がひどい場合は避ける必要があります。使用中に異常を感じたときはすぐに使用を中止し、医師に相談しましょう。
ケトクリームは毎日決まった回数で使うことが大切です。使ったり使わなかったりすると、効果が十分に発揮されず、治療に時間がかかる可能性があります。忘れずにコツコツ続けることで、しっかり治しましょう。
まれにかゆみや赤み、刺激感などの副作用が出ることがあります。これは皮膚が薬に反応するために起こるもので、多くの場合は軽い症状にとどまります。異常を感じたら、すぐに使用をやめて医師に相談してください。
妊娠中は、治療の必要性とリスクをよく考えたうえで、医師の判断のもと使用することが必要です。ケトクリームは皮膚からの吸収がほとんどないとされていますが、妊娠中は慎重な対応が求められます。
ケトクリームを使ったときに、塗った部分がヒリヒリしたり赤くなることがあります。これは副作用の一つとして知られており、多くは軽症ですが、ひどい場合は使用を中止して医師に相談するのが安心です。
ケトクリームを必要以上にたくさん使っても効果が高まるわけではなく、むしろ皮膚に刺激が出やすくなることがあります。適量を守って使うことが、副作用を防ぐポイントになります。
| 1日の使用回数 | 1回 |
|---|---|
| 1回の使用量 | 適量 |
| 使用のタイミング | 入浴後など患部を清潔にしたとき |
| 使用間隔 | 24時間以上 |
| 使用する箇所 | 症状のある箇所 |
| 商品名 | クロトリマゾールクリーム | クロトリマゾールローション | ルリコンクリーム・ジェネリック | ユーモゾンM | トピカゾールクリーム | マックダームK10クリーム | 十足美 |
|---|---|---|---|---|---|---|---|
| 商品画像 |  |  |  |  |  |  | |
| 特徴1 | ・全身性の副作用の心配が少ない | ・ベタつきにくく使いやすいローションタイプ | ・カンジダや水虫など幅広い症状に有効 | ・2つの有効成分が速やかに症状を鎮静 | ・患部にさっと塗って使用できる | 患部にピンポイントに使えるクリームタイプ | ・角質を溶かして水虫を撃退 |
| 特徴2 | ・強い抗菌活性を持つ有効成分を配合 | ・人間の細胞には悪影響を与えない | ・クリームタイプで使いやすい | ・外用薬で副作用はほとんどない | ・副作用の心配がほとんどなく、安全性が高い | 複数の有効成分が水虫と二次感染を改善 | ・副作用が少なく安心して使用できる |
| 内容量 | 1%30gx1本 | 1%30mlx1本 | 10gx1本 | 15gx1本 | 1%30gx1本 | 15gx1本 | 1箱 |
| 価格 | 2,560円 | 3,760円 | 3,360円 | 4,260円 | 3,560円 | 4,160円 | 1,750円 |
| 0.1〜5%未満 | 0.1%未満 | 頻度不明 | |
| 皮膚 | 接触皮膚炎、そう痒、発赤、刺激感、紅斑、糜爛、皮膚剥脱 | 水疱、亀裂、疼痛、皮膚灼熱感、発疹、皮膚のべとつき感 | 蕁麻疹 |
| 全身障害及び 投与局所様態 | 適用部位反応(乾燥、浮腫) | 適用部位反応(出血、不快感、炎症、錯感覚) | |
| 免疫系障害 | 過敏症 |
本製品は海外製のため、期限表記が日本と異なる場合がございます。
パッケージ裏面や側面、シートなどに以下のような表記がされています。
| EXP | 使用期限 例:EXP 12/2025→2025年12月まで使用可 |
|---|---|
| MFG または MFD | 製造日 例:MFG 03/2023 |
| BEST BEFORE | 品質が最も安定している目安日 |
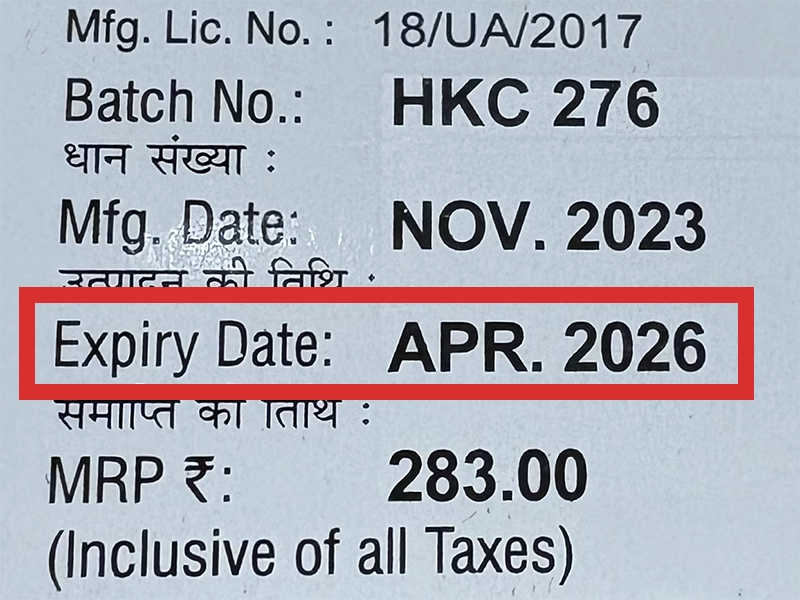
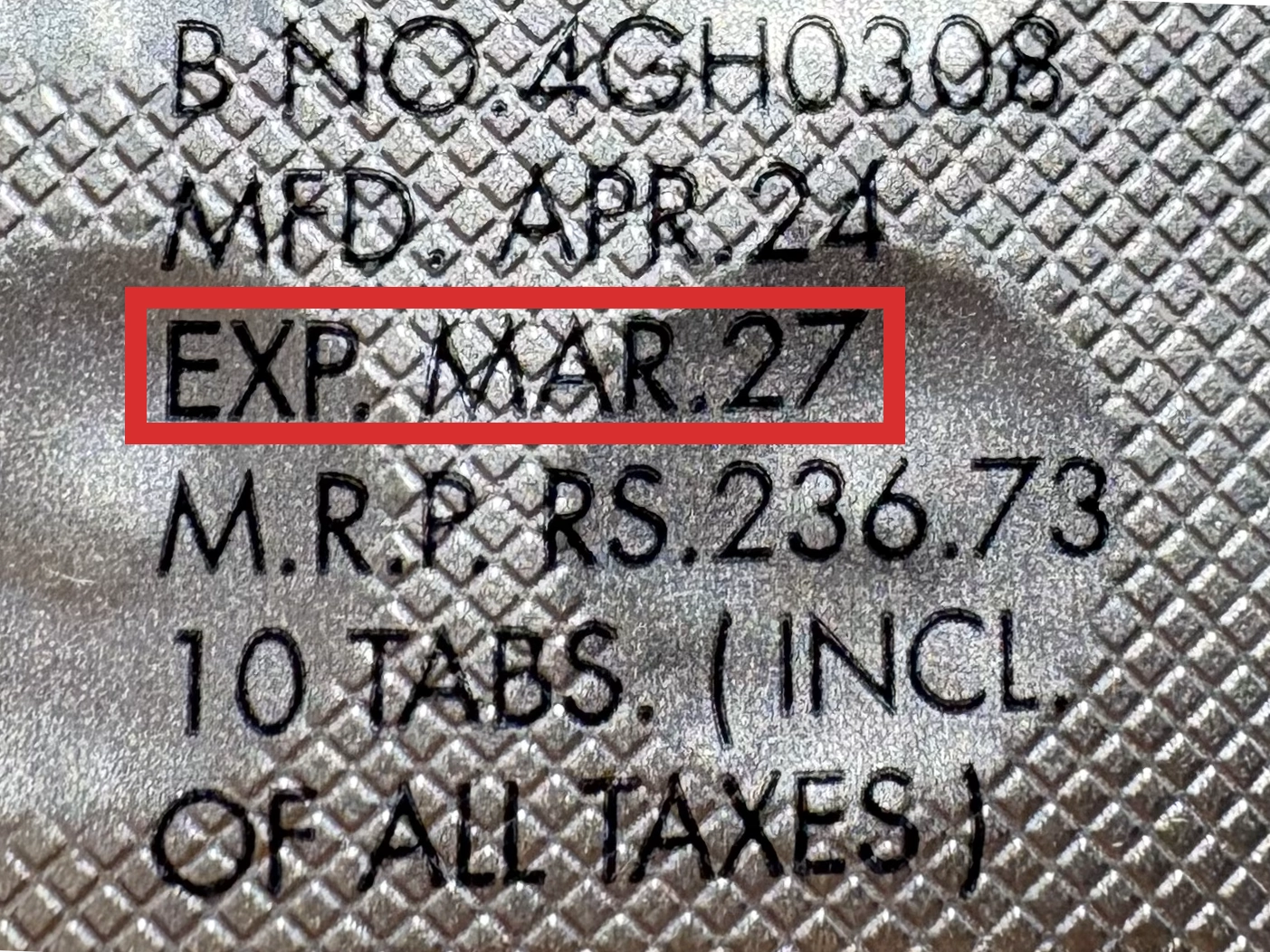
※国や製品により日付の並び(例:月/年、日/月/年)が異なる場合がありますのでご注意ください
EXP(Expiry Date) の表記がなく、MFG または MFDしか記載がないケースがあります。
この場合は MFG(MFD) から2~3年が使用期限の目安です。
※「LOT」や「BATCH」の表記は製造番号であり期限ではありません。

パッケージ例となります。
商品やご注文単位によってはシート単位でのお届けとなる場合が御座います。
外箱に当サイト名や商品名が記載されることはないため、ご家族や配達員など第三者に内容を知られることは御座いません。
カンジダになったとき、何度か使ったことがありますが、綺麗に治るかわりにやや副作用が出る。一瞬カンジダが悪化したのかと不安になってしまいました。
23歳女ですが、水虫です・・・。誰にも言えないくらい恥ずかしく、でもそのままにもしておきたくないのでこれを買いました。はじめて局留めで受け取りましたが、難しくなかったです。使って3日になりますが、状態は良くなってて、痒さはほとんどありません。
生理前後にカンジダになることが多いため、使用してます。そろそろ生理きそうだなってときはポーチへ入れるようにしてますので、あんまりムズムズしません。今後も1本はストックしておきたい薬です
前から悩んでた背中のポツポツがこちらのクリームで治りました!!何を塗ってもダメだったから半ばあきらめてましたが治ってくれて本当にヨカッタ!!!チャレンジしてみるものですね(^^)
水虫治療のため、毎日入浴後に足の指の間に塗り込んでます。痒み自体は塗ればすぐに軽減されますが、菌の死滅は難しいようでなかなか治りません。
商品口コミの投稿は会員のみ行えるようになっております。
お手数ですが会員ログインの上でご投稿頂きますようお願いいたします。
口コミをご投稿頂いたお客様にはポイントをプレゼントさせて頂いております。
文章のみであれば100ポイント、文章+写真付きのものは300ポイントをプレゼントさせて頂きます。
規約や詳細などはこちらをご確認くださいませ。